-
Production of Quicklime (CaO) — The Most Critical Industrial Application
-
Construction Industry: Reacts with water to form slaked lime (Ca(OH)₂), which is used in masonry mortar, plastering, and lime putty coatings.
-
Iron and Steel Smelting: Serves as a flux to remove impurities like sulfur and phosphorus from molten iron.
-
Flue Gas Desulfurization: Utilized in coal-fired power plants to remove sulfur dioxide (SO₂).
-
Chemical Raw Material: Applied in the manufacturing of bleaching powder, calcium carbide (CaC₂), sodium hydroxide, etc.✅ Without calcination, it is impossible to obtain highly reactive CaO from natural limestone.
-
Release of High-Purity Carbon Dioxide (CO₂)
-
In certain industrial scenarios (e.g., production of food-grade CO₂, dry ice manufacturing, greenhouse gas fertilization), limestone calcination is one of the sources for obtaining pure CO₂.
-
The CO₂ generated from the reaction can be purified and used in beverage carbonation, welding shielding gas, etc.
-
-
Modification of Physicochemical Properties to Meet Specific Application Requirements
-
Calcined CaO exhibits high reactivity, strong alkalinity, and excellent water absorption, whereas raw CaCO₃ is chemically inert and insoluble in water.
-
Examples:
-
In soil improvement, CaO is used to rapidly increase soil pH.
-
In desiccants, the exothermic and water-absorbing properties of the reaction CaO+H₂O→Ca(OH)₂ are harnessed.
-
-
-
Precursor Step for Preparing Special Materials
-
-
In the production of calcium carbide (CaC₂), limestone must first be calcined into CaO, which then reacts with coke at high temperatures in an electric furnace:
-
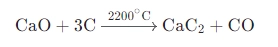
-
Calcium carbide is used in acetylene gas production and plays an important role in welding and chemical synthesis.
-
-
CaCO₃ cannot rapidly neutralize acidic wastewater.
-
It does not react vigorously with water to release heat.
-
It cannot effectively participate in the slagging process during steelmaking.
|
Parameter
|
Requirements
|
Rationale
|
|
Temperature
|
900–1200°C
|
Insufficient temperature leads to incomplete reaction; excessively high temperature causes “over-burning” of CaO (densification and reduced reactivity).
|
|
Time
|
Sufficient residence time
|
Ensures complete decomposition of the internal structure of particles.
|
|
Raw Material Particle Size
|
Moderate (typically 20–50 mm)
|
Oversized particles result in slow heat transfer; undersized particles are prone to being carried away by airflow.
|
-
Calcination is a high-energy-consumption and high-carbon-emission process (approximately 0.8 tons of CO₂ are emitted per ton of CaO produced).
-
Approximately 7% of global anthropogenic CO₂ emissions stem from cement and lime production (with lime calcination accounting for a considerable proportion).
-
Therefore, the industry is exploring the following solutions:
-
Energy-saving technologies such as oxygen-enriched combustion and electric kilns.
-
Carbon dioxide capture, utilization, and storage (CCUS).
-
Alternative cementitious materials (e.g., geopolymers).
-