1. Pharmacopoeia Mandatory Rules
2. Industry-Standard Brightness Specifications (Commercial Pharmaceutical Grade)
| Grade | Minimum Brightness Requirement | Typical Brightness Range | Application Scenarios |
| Entry-level Compliant Pharmaceutical Grade | ≥ 93.0% | 93.0-95.0% | Meets basic pharmacopoeia requirements, for generic oral solid preparations, excipient fillers with low appearance requirements |
| Mainstream Commercial Pharmaceutical Grade (USP/EP/BP Compliant) | ≥ 95.0% | 95.0-97.0% | Global pharma industry standard, for tablets, capsules, antacids, calcium supplements, and most common pharmaceutical formulations; the most widely used specification in the market |
| High-End Premium Pharmaceutical Grade | ≥ 97.0% | 97.0-98.5% | For high-end formulations with strict appearance and color consistency requirements, such as chewable tablets, effervescent tablets, orally disintegrating tablets, and topical pharmaceutical creams |
3. Key Notes
- The brightness of pharmaceutical CaCO₃ is directly determined by the content of color-causing impurities (Fe, Mn, Ti, etc.). Qualified pharmaceutical products usually control iron content below 50 ppm (far lower than the pharmacopoeia limit of 200 ppm), which naturally ensures a brightness of over 93%.
-
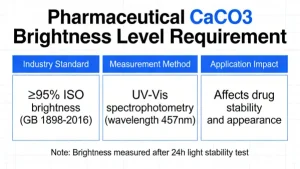
For pharmaceutical excipient use, most regional drug regulatory authorities and pharmaceutical manufacturers adopt an internal control brightness standard of≥ 95.0% (ISO R457)as the qualified baseline for incoming material inspection.