
Why Does Calcium Carbonate Absorb Heat but Not Water?
The fact that calcium carbonate absorbs heat is determined by its thermodynamic characteristics and chemical...

Why Is Calcium Carbonate Added to Food?
Calcium carbonate is added to food primarily to fortify nutrition, adjust texture and stability, and...

Why is calcium carbonate yellow?
Pure calcium carbonate (CaCO₃) is inherently white or colorless and does not appear yellow. If...
Why Does the Human Body Need Calcium Carbonate?
The human body does not directly require calcium carbonate (CaCO₃) as a compound; rather, it...
Why do calcium carbonate granules contain sugar?
Calcium Carbonate (CaCO₃) is an inorganic compound that contains no sugar in its chemical structure....

Why Does Calcium Carbonate Turn Yellow When Mixed with Water?
Calcium carbonate (CaCO₃) itself is a white, odorless solid that is insoluble in water. It...
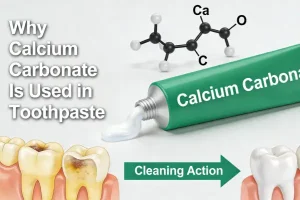
Why Calcium Carbonate Is Used in Toothpaste
The addition of calcium carbonate (CaCO₃) in toothpaste is a common practice in the industry....

Why Calcium Carbonate Is Used in Iron Smelting?
In the iron smelting process, calcium carbonate (usually added in the form of natural minerals...

Why Calcium Carbonate Is Used in PVC?
Calcium carbonate is widely used in the processing of PVC (polyvinyl chloride). The core reason...

How to Judge Whether Calcium Carbonate Has Undergone Hydrolysis?
The core logic for judging whether calcium carbonate has undergone hydrolysis is: focus on theprotontransfer...

Under What Circumstances Does Calcium Carbonate Undergo Hydrolysis?
Based on the chemical properties of calcium carbonate, calcium carbonate hardly undergoes observable hydrolysis in...

Why Doesn’t Calcium Carbonate Undergo Hydrolysis?
Calcium carbonate (CaCO₃) does not undergo hydrolysis reactions. The core reason lies in the ionization...
