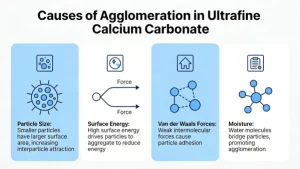
Agglomeration in ultrafine calcium carbonate (UFCC)—typically particles <1 µm, often in the 40–200 nm range—is a common challenge that affects dispersion, flowability, and performance in end-use applications (e.g., plastics, coatings, paper, pharmaceuticals). This phenomenon arises from several interrelated physical and chemical factors:
High Surface Energy
Ultrafine particles have very high specific surface area, leading to high surface energy.
To reduce this energy, particles tend to aggregate or agglomerate spontaneously.
This is driven by thermodynamics: agglomeration lowers the total surface free energy of the system.
Van der Waals Forces
Dominant at nanoscale distances (<100 nm).
These attractive interparticle forces are strong relative to particle mass, making separation difficult without external energy or surface modification.
Moisture and Capillary Forces
Even small amounts of adsorbed water can form liquid bridges between particles.
Capillary forces from moisture significantly increase agglomeration, especially in humid environments or during wet processing followed by incomplete drying.
Electrostatic Interactions
In dry systems, triboelectric charging during grinding or handling can cause particles to attract or repel.
In wet systems, low zeta potential (near the isoelectric point of CaCO₃, ~pH 8–9) reduces electrostatic repulsion, promoting agglomeration.
Lack of Surface Modification
Uncoated UFCC has a hydrophilic, polar surface that favors particle-particle interaction.
Without surface treatment (e.g., stearic acid, titanates, silanes), particles remain prone to agglomeration.
Processing Conditions
Drying methods: Spray drying or oven drying can fuse particles if not controlled.
Mechanical compaction: Storage or pneumatic conveying under pressure can form hard agglomerates.
Rapid precipitation during synthesis can trap particles together before stabilization.
Crystal Structure and Morphology
Certain crystal habits (e.g., rhombohedral calcite vs. needle-like aragonite) may pack more tightly or interlock, enhancing agglomeration.
Irregular shapes increase contact points and friction.
Strategies to Reduce Agglomeration
| Approach | Mechanism |
| Surface coating (e.g., fatty acids like stearic acid) | Creates steric hindrance and makes surface hydrophobic |
| Controlled drying (e.g., freeze drying, fluidized bed) | Minimizes liquid bridge formation |
| Dispersion aids / surfactants | Improve wetting and electrostatic/steric stabilization |
| Deagglomeration equipment (e.g., jet mills, high-shear mixers) | Breaks soft agglomerates post-synthesis |
| Storage in dry, inert atmosphere | Prevents moisture uptake and caking |
Agglomeration in ultrafine calcium carbonate is primarily driven by high surface energy and interparticle forces, exacerbated by moisture and lack of surface passivation. Effective mitigation requires a combination of surface chemistry control, optimized processing, and proper handling/storage.
For industrial applications, surface-treated UFCC with controlled moisture (<0.5%) and narrow particle size distribution typically exhibits the best dispersibility and minimal agglomeration.