Removing silica (SiO₂) from limestone (predominantly CaCO₃) before grinding is a critical beneficiation step to improve the purity of calcium carbonate products, reduce wear on grinding equipment, and eliminate silica-related impurities in downstream applications (e.g., paper, plastics, construction). The optimal method depends on two key ore characteristics: the occurrence state of silica (free silica/quartz vs. combined silica like silicates) and its dissemination grain size, as well as the required CaCO₃ purity (industrial grade vs. high-purity food/pharmaceutical grade).
Silica in limestone typically exists in three forms:
Free silica: Discrete quartz particles or siliceous gangue (e.g., sand) with coarse/fine dissemination.
Surface siliceous slime: Fine silica mud adhering to limestone particle surfaces.
Combined silica: Silicates (e.g., feldspar, clay minerals) or silicoaluminates embedded in the limestone matrix (hard to separate via physical methods).
Below are the industrial-scale, cost-effective methods for silica removal before grinding, sorted by popularity (physical/physicochemical methods first, chemical methods for high-purity requirements), with detailed principles, processes, and application scenarios:
Core Physical & Physicochemical Methods (Mainstream for Industrial Use)
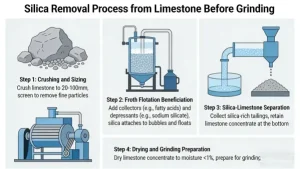
These methods are preferred for most limestone beneficiation due to low cost, high efficiency, and no damage to CaCO₃ crystals, and are suitable for removing free silica and surface siliceous slime (the most common silica forms in limestone). All methods require a pre-crushing & screening step first: crush limestone to a suitable particle size (0–5 mm, matching the beneficiation equipment) and screen into different size fractions for classified treatment (improves separation efficiency).
1. Washing & Scrubbing (Simplest, for Surface Slime/Coarse Free Silica)
Principle
Utilize mechanical scrubbing and hydraulic classification to separate loose siliceous slime (≤0.074 mm) adhering to limestone surfaces, or coarse free silica (quartz/sand, ≥0.5 mm) with obvious density/particle size differences from limestone.
Process
Scrubbing: Use a cylindrical scrubber, trough scrubber, or high-shear scrubber to agitate limestone particles with water (solid-liquid ratio 1:1–1:3) – mechanical friction peels off surface siliceous slime.
Hydraulic separation: Pass the scrubbed slurry through a spiral classifier, hydrocyclone, or vibrating screen – water washes away light siliceous slime, and coarse quartz sand is screened out by particle size.
Application
Suitable for limestone with low silica content (<3%) and silica mainly in the form of surface slime/coarse free silica (e.g., limestone for construction cement).
Advantages
Low investment/operating cost, simple process, no chemical reagents, environmental friendliness.
2. Flotation (Most Efficient, for Fine Disseminated Free Silica)
The most widely used method for silica removal in industrial CaCO₃ production, as limestone (calcite/aragonite) and quartz have very similar densities (CaCO₃: 2.7 g/cm³; SiO₂: 2.65 g/cm³) – physical methods (e.g., gravity separation) are ineffective for fine disseminated silica (0.01–0.5 mm).
Core Type: Reverse Flotation (Preferred for Limestone)
Float and separate silica gangue while inhibiting limestone (opposite to direct flotation, which floats limestone – reverse flotation has higher selectivity and CaCO₃ recovery).
Key Process Parameters (Industrial Standard)
| Reagent Type | Common Reagents | Function & Dosage |
| pH Regulator | NaOH, CaO | Adjust slurry pH to 10–12 (alkaline environment optimizes silica inhibition/collection). |
| Silica Depressant | Sodium silicate (water glass), sodium hexametaphosphate | Inhibit limestone flotation, adsorb on silica surfaces to enhance hydrophilicity (dosage: 500–1500 g/t ore). |
| Silica Collector | Fatty acids (oleic acid, stearic acid), fatty acid soaps | Hydrophobic modification of silica particles for bubble attachment (dosage: 100–300 g/t ore). |
| Frother | Pine oil, terpineol (optional) | Stabilize flotation bubbles (for fine silica). |
Equipment
Mechanical agitation flotation machine (for coarse slime, 0.074–0.5 mm) or column flotation machine (for fine slime, ≤0.074 mm) – multi-stage flotation (roughing + cleaning) for higher silica removal efficiency.
Application
Suitable for limestone with medium-high silica content (3–10%) and fine disseminated free silica (the main method for producing industrial-grade CaCO₃ for coatings/paper).
Advantages
High silica removal rate (can reduce SiO₂ content to <0.5%), high CaCO₃ recovery (>90%); adjustable process for different purity requirements.
3. Gravity Separation (Auxiliary, for Coarse Disseminated Silica)
Principle
Utilize minor density differences between limestone and silica, plus gravity/centrifugal force, to separate coarse disseminated silica (≥0.1 mm) – only an auxiliary method (density difference is too small for fine silica).
Common Processes
Jigging: Use a jig machine for coarse particles (≥0.5 mm) – suitable for limestone with silica in the form of massive quartz gangue.
Shaking table: For medium particles (0.1–0.5 mm) – separate silica from limestone in the presence of other heavy gangue (e.g., iron ore).
Heavy medium separation: Use a dense medium (e.g., magnetite slurry) to magnify density differences – for large limestone lumps (5–50 mm) with coarse silica veins.
Application
Combined with washing/flotation for limestone with mixed coarse/fine silica – pre-remove coarse silica to reduce subsequent flotation load.
4. Magnetic Separation (Specialized, for Silica with Magnetic Impurities)
Most silica (quartz) is non-magnetic, so this method is only effective when silica is associated with magnetic gangue (e.g., magnetite, hematite) or modified via magnetic seed coating (physicochemical modification).
Core Process
Use a high-gradient magnetic separator (HGMS) or high-intensity magnetic separator (10.000–20.000 Oe) to separate magnetic siliceous gangue from non-magnetic limestone.
Magnetic seed coating: Adsorb magnetic particles (e.g., Fe₃O₄) on silica surfaces to make it magnetic, then separate via low-intensity magnetic separation (for fine non-magnetic silica).
Application
Suitable for limestone with silica associated with magnetic impurities (e.g., iron-bearing silicates) – usually combined with flotation for deep silica removal.
Chemical Methods (For High-Purity CaCO₃ & Combined Silica)
Chemical methods are used only when limestone contains combined silica (silicates/silicoaluminates) (physical methods cannot separate embedded silica) or when producing high-purity CaCO₃ (food/pharmaceutical/electronic grade, SiO₂ <0.1%). These methods have higher cost and require wastewater treatment but achieve ultra-deep silica removal.
Core Method: Alkaline Leaching (NaOH Leaching)
Principle
Silica (free/combined) reacts with concentrated NaOH at high temperature to form water-soluble sodium silicate (Na₂SiO₃, water glass), which is then removed by water washing; limestone (CaCO₃) is inert to NaOH under controlled conditions (avoids formation of insoluble Ca(OH)₂).
Industrial Process Conditions
Crush limestone to fine powder (0.074–0.15 mm) (grinding to fine size increases reaction contact area – *mild pre-grinding, not the final product grinding*).
Mix with 10–20% NaOH solution (solid-liquid ratio 1:4–1:6) and heat to 80–100℃ (reflux to prevent NaOH volatilization).
Stir for 1–3 h – SiO₂ + 2NaOH = Na₂SiO₃ + H₂O (combined silicates also react to form soluble silicates).
Solid-liquid separation: Filter the slurry to separate CaCO₃ solids from Na₂SiO₃-containing lye.
Washing & neutralization: Wash CaCO₃ solids with deionized water 3–5 times to remove residual Na₂SiO₃/NaOH; neutralize residual alkali with dilute HCl (pH to 7–8) if needed.
Key Control Points
Avoid excessive NaOH concentration/temperature (prevents CaCO₃ from reacting with NaOH to form Ca(OH)₂, which precipitates and reduces CaCO₃ purity).
Recycle NaOH lye (concentrate and reuse) to reduce cost and environmental pollution.
Acid Leaching (Limited Application)
Acid leaching (dilute HCl/H₂SO₄) is rarely used alone because CaCO₃ reacts vigorously with acids (CaCO₃ + 2HCl = CaCl₂ + CO₂↑ + H₂O), which dissolves the target limestone. It is only used as a post-treatment step (after alkaline leaching) to remove trace calcium hydroxide/silicate impurities for ultra-high-purity CaCO₃.
Combined Beneficiation Processes (For Complex Limestone Ores)
Most limestone ores contain silica in mixed forms (surface slime + fine disseminated free silica + minor combined silica). A combined physical-chemical process is the most economical and efficient solution, as it reduces the load of high-cost flotation/chemical leaching by pre-removing simple silica.
Common Industrial Combined Processes
Washing → Scrubbing → Flotation (Reverse): For limestone with surface siliceous slime + fine disseminated free silica (the most widely used combined process for industrial-grade CaCO₃).
Crushing → Screening → Gravity Separation → Flotation: For limestone with coarse quartz gangue + fine silica (pre-remove coarse silica via gravity separation to cut flotation reagent consumption).
Washing → Flotation → Alkaline Leaching: For limestone with medium silica content + minor combined silica (produces high-purity CaCO₃ for coatings/paper).
Scrubbing → Magnetic Separation → Flotation: For limestone with silica associated with magnetic impurities (deep silica removal).
Key Industrial Considerations for Pre-Grinding Silica Removal
Ore Property Analysis First: Conduct XRD, rock phase analysis, and particle size analysis to determine silica’s occurrence state, dissemination grain size, and content – the foundation for method selection (avoid blind process design).
Cost Priority: Follow the principle “physical methods first → flotation → chemical methods”; chemical methods are only for high-purity requirements.
Reagent/ Wastewater Treatment: Recycle flotation reagents (e.g., fatty acid collectors) and alkaline leaching lye (NaOH) to reduce cost; treat flotation wastewater (coagulation + sedimentation) and chemical leaching wastewater (neutralization + desalination) to meet environmental discharge standards.
Equipment Matching: Select beneficiation equipment based on ore processing capacity and particle size (e.g., hydrocyclones for fine slime, column flotation machines for ultra-fine silica).
Grinding Synergy: The beneficiated limestone (low silica) is sent to grinding – lower silica content significantly reduces wear on ball mills/roller mills and extends equipment service life.
Summary of Method Selection
| Silica Form & Content | Recommended Method | Target SiO₂ Content | Application Grade |
| Surface slime/coarse free Si, <3% | Washing + Scrubbing | <1% | Construction/cement |
| Fine disseminated free Si, 3–10% | Reverse Flotation (multi-stage) | <0.5% | Industrial coatings/paper |
| Mixed coarse/fine free Si, 5–12% | Gravity Separation + Flotation | <0.3% | High-grade coatings |
| Combined silica + free Si, >8% | Flotation + Alkaline Leaching | <0.1% | Food/Pharmaceutical/Electronic |